ONCOLYTIC VIRUS INDUCED PYROPTOSIS AS A NOVEL CANCER IMMUNOTHERAPY
2023-01-17
Inducing tumor-reactive T cells against cancer recurrence
PD-1 immune checkpoint blockade (ICB) transforms clinical cancer management, but adaptive resistence frequently occurs. PD-1 monotherapy generally fails to induce tumor-reactive memory CD8+ T cells, which may primarily accounting for high recurrence rates post PD-1 ICB.
We are developing a PD-1-ICB-based combo immunotherapy by targeting tumor cell pyroptosis. Such combination effectively induces the generation of tumor-reactive pan-tissue circulating memory CD8+ T (ptc-Tmem) cells.
The presence of ptc-Tmem cells prolong the anti-tumor effects by preventing local or distal tumor recurrence.
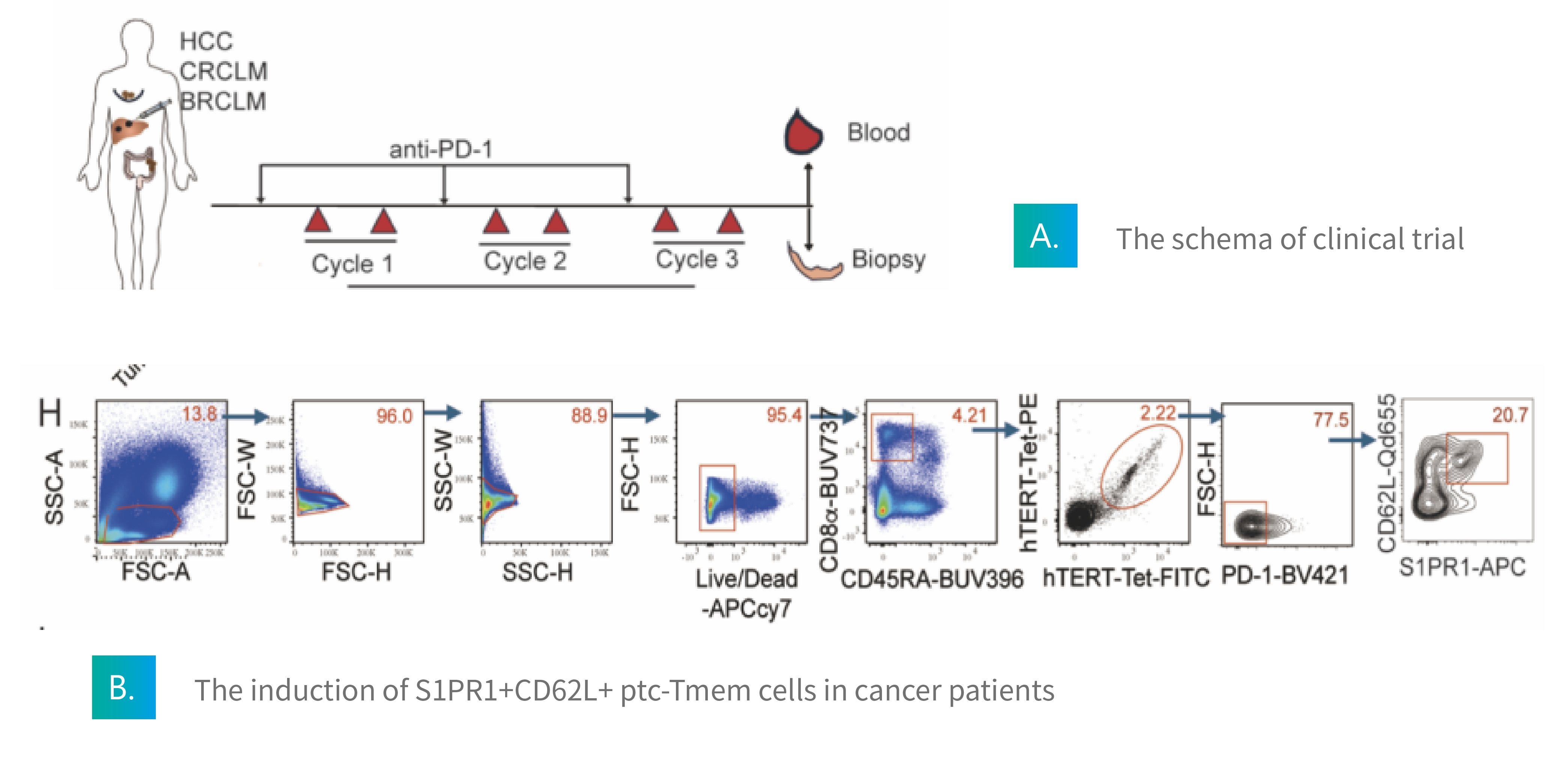
The ongoing clinical trial (NCT 07018518) of this como immunotherapy achieves encouraging results in hepatocellular cacinoma (HCC) patients.
Oncolytic virus induced pyroptosis combined with PD-1 ICB generates pan-tissue circulating memory T (ptc-Tmem) cells
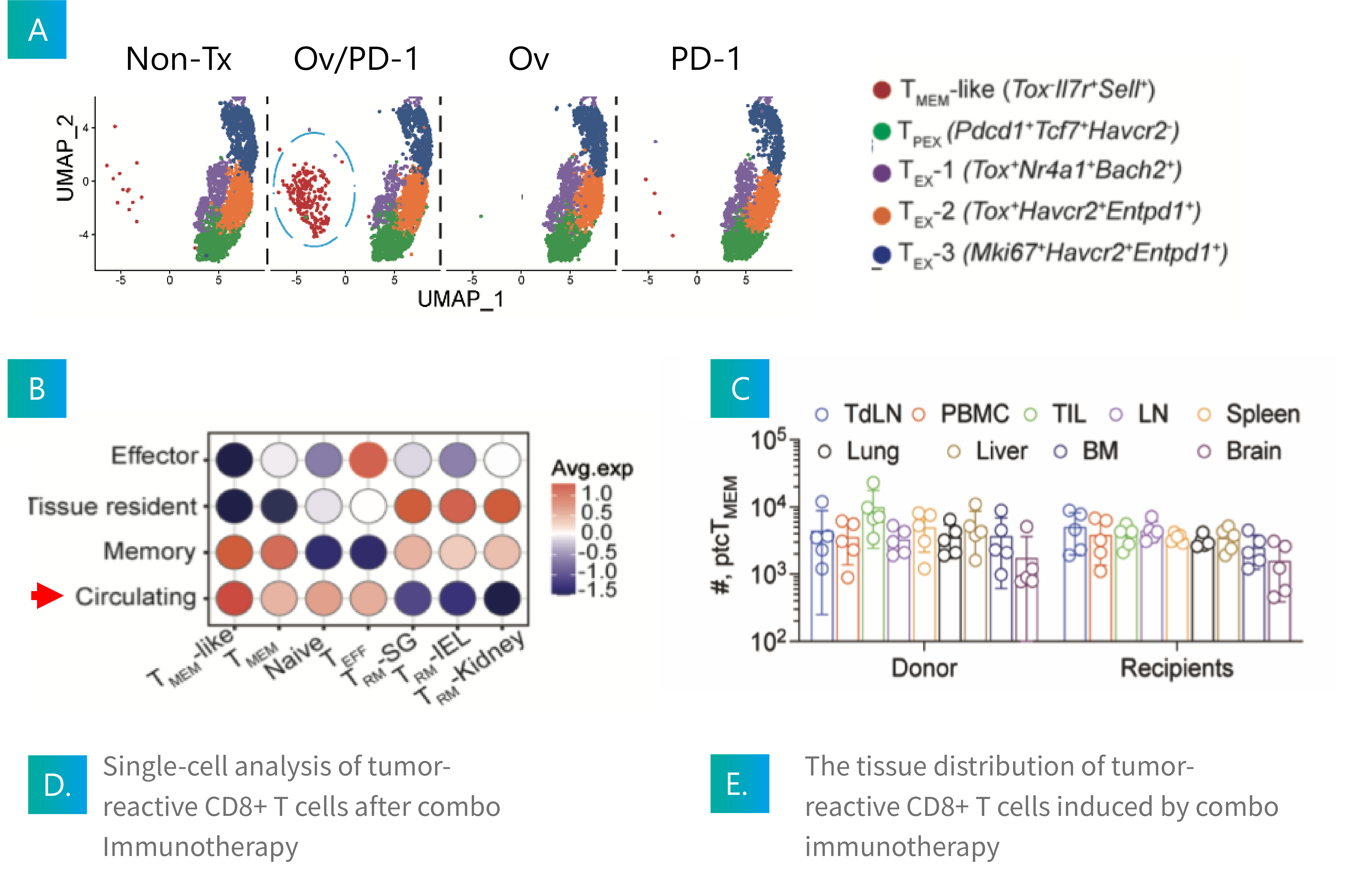
The induction of ptc-Tmem cells are necessary and sufficient for long-term tumor control
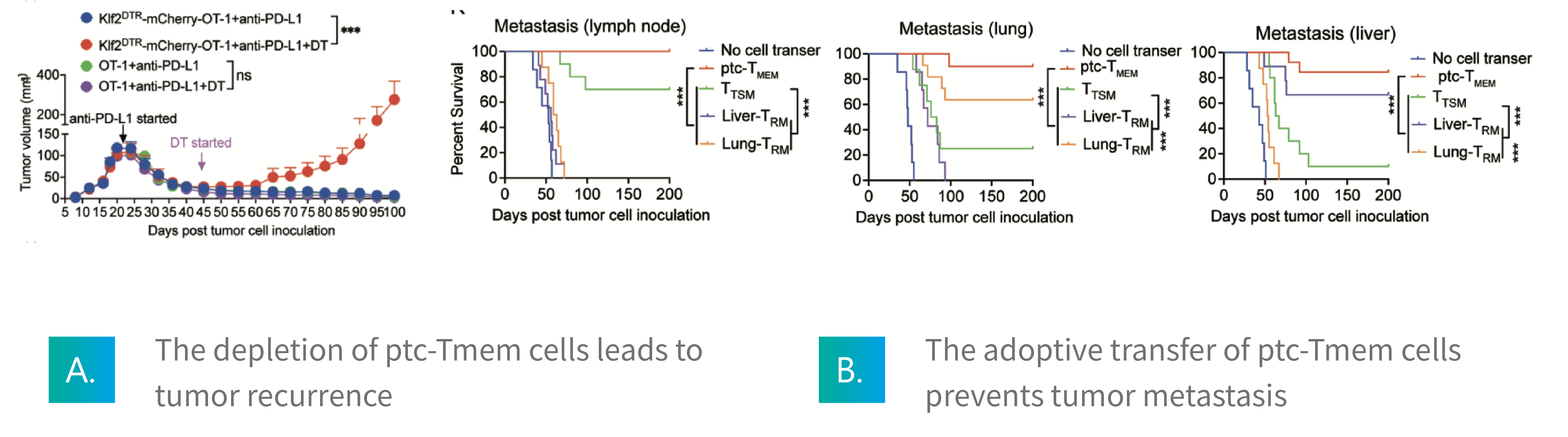
Oncolytic virus induced pyroptosis combined with PD-1 ICB generates pan-tissue circulating memory T (ptc-Tmem) cells
A NOVEL ANTI-LIVER CANCER IMMUNOTHERAPY STRATEGY BASED ON PYROPTOSIS
China bears the highest global burden of new liver cancer cases and related mortality. The five-year survival rate for patients with advanced hepatocellular carcinoma remains as low as 14.4%, and the objective response rate to existing immunotherapies is less than 15%. Notably, in contrast to Western countries where hepatitis C virus and metabolic factors predominate, approximately 80% of liver cancer cases in China are attributable to chronic hepatitis B virus (HBV) infection. Due to this divergence, funding and research investment from Europe and the United States in this specific area remain limited. Effectively controlling HBV-induced liver cancer constitutes a critical priority for China's "Healthy China" strategy.
In response to this critical national health priority, the Cancer and Immunotherapy Team at Changping Laboratory has integrated their pioneering research on "tumor cell pyroptosis" and "T-cell exhaustion and memory"—including the discovery of the novel pyroptosis mediator GSDMB (2020, Science; 2020, Nature) and new mechanisms for generating memory T cells under conditions of immune exhaustion (2016, Nature; 2022, Cell). Leveraging these advances, they engineered an oncolytic virus to deliver the GSDMB target, enabling highly selective infection of cancer cells and guiding T lymphocytes to specifically recognize infected tumor cells and release granzyme A, a protease capable of cleaving GSDMB. Through this dual-targeting strategy, tumor cells are highly specifically induced to undergo pyroptosis, leading to robust release of tumor antigens and inflammatory cytokines. This process promotes the generation of increased numbers of memory T cells in tumor-draining lymph nodes, which subsequently migrate to tumor sites and efficiently eliminate malignant cells. Importantly, following effective control of tumor burden, anti-tumor memory T cells persist long-term and surveil multiple organs—including common metastatic sites such as the lung, liver, lymph nodes, brain, and bone—thereby providing durable suppression of tumor recurrence and metastasis.
The pyroptosis-inducing oncolytic virus immunotherapy (hereinafter referred to as PIN therapy) is distinct from oncolytic viruses developed in Europe and the United States in both molecular target and mechanism of action. Those conventional agents primarily depend on the oncolytic ability of the viruses, elicit limited immune activation, and fail to generate durable immune memory. Consequently, the clinical efficacy of approved oncolytic virus therapies such as T-Vec remains suboptimal. To date, PIN therapy has been evaluated in a cohort of 11 patients with advanced hepatocellular carcinoma who progressed after standard-of-care treatments. Of these, seven patients achieved disease control (objective response rate: 63.6%), including three who attained complete remission (complete remission rate: 27.2%). These outcomes are markedly superior to those associated with current international standard-of-care regimens, which typically yield an objective response rate of approximately 20% and a complete remission rate below 5%. The research team is actively advancing further clinical development of PIN therapy for advanced liver cancer.
PIN therapy is an original and innovative immunotherapeutic strategy independently developed by Changping Laboratory, protected by fully owned intellectual property rights. Based on current clinical evidence, PIN therapy has the potential to emerge as a safe and highly effective treatment for advanced hepatocellular carcinoma, significantly prolonging overall survival, potentially achieving clinical cure in some patients, and providing a crucial clinical solution to improve China's five-year survival rate for hepatocellular carcinoma.